DUKE TODAY - DURHAM, N.C. – Magnetic resonance imaging (MRI) is how we visualize soft, watery tissue that is hard… Read More
Small Animal Imaging Laboratory (SAIL)
CIVM, in collaboration with the Duke Pratt School of Engineering, provides state of the art imaging services for preclinical and basic science studies for the entire university. Featured equipment includes the 7T Bruker MRI System and the MILabs MicroSPECT System. Learn More

Duke Imaging Innovation Laboratory (DIIL)
With DIIL, CIVM is developing the next generation of imaging tools, leveraging the engineering expertise at Duke to push the technical boundaries of multimodal imaging. Featured equipment includes the 7 T Agilent MRI System and the 9.4T MRI System System. Learn More

Request a Project Collaboration
All project requests start here with your review of CIVM resources and rates and your submission of our online Project Request Form, either for SAIL or for DIIL. You'll be asked to provide the details of your project, including imaging needs, specific aims, timing and funding. Learn more and begin the project request form by choosing either SAIL or DIIL.
Recent Publications
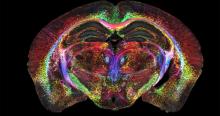
Johnson GA, Tian Y, Ashbrook DG, Cofer GP, Cook JJ, Gee JC, Hall A, Hornburg K, Kaczorowski CC, Qi Y, Yeh FC, Wang N, White LE, Williams RW. Merged magnetic resonance and light sheet microscopy of the whole mouse brain. Proc Natl Acad Sci U S A. 2023 Apr 25;120(17):e2218617120. doi: https://www.pnas.org/doi/10.1073/pnas.2218617120 Epub 2023 Apr 17. Erratum in: Proc Natl Acad Sci U S A. 2023 Jun 20;120(25):e2308718120. PMID: 37068254; PMCID: PMC10151475.
Blocker, S. J., Morrison, S., Everitt, J. I., Cook, J., Luo, S., Watts, T. L., & Mowery, Y. M. (2023). Whole-Slide Cytometric Feature Mapping for Distinguishing Tumor Genomic Subtypes in Head and Neck Squamous Cell Carcinoma Whole-Slide Images. Am J Pathol, 193(2), 182–190. https://doi.org/10.1016/j.ajpath.2022.11.004
Hornburg, K. J., Slosky, L. M., Cofer, G., Cook, J., Qi, Y., Porkka, F., Clark, N. B., Pires, A., Petrella, J. R., White, L. E., Wetsel, W. C., Barak, L., Caron, M. G., Johnson, G. A. (2023). Prenatal heroin exposure alters brain morphology and connectivity in adolescent mice. Nmr Biomed, 36(2), e4842. https://doi.org/10.1002/nbm.4842
Enam SF, Kilic CY, Huang J, Kang BJ, Chen R, Tribble CS, Ilich E, Betancur MI, Blocker SJ, Owen SJ, Buckley AF, Lyon JG, Bellamkonda RV. Cytostatic hypothermia and its impact on glioblastoma and survival. Sci Adv. 2022 Nov 25;8(47):eabq4882. https://doi.org/10.1126/sciadv.abq4882